Galvanizing Process
The galvanizing process consists of three basic elements: surface preparation, galvanizing, and inspection.
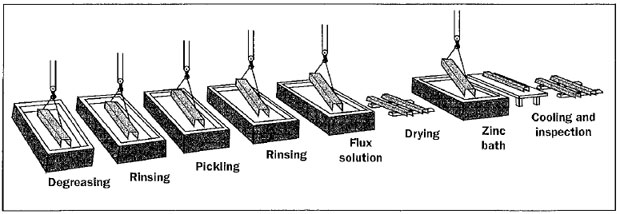
PHOTO: American Galvanizers Association
Surface Preparation
Surface preparation is the most important step in the application of any coating. In most instances where a coating fails before the end of its expected service life, it is because of incorrect or inadequate surface preparation.
The surface preparation step in the galvanizing process has its own built-in means of quality control because zinc simply will not react with a steel surface that is not perfectly clean.
Any failures or inadequacies in surface preparation will be immediately apparent when the steel is withdrawn from the liquid zinc because the unclean areas will remain uncoated, and thus can be corrected.
On-site painting or other field-applied systems of corrosion protection may involve the use of different subcontractors and/or work groups to prepare the surface and apply the coating. This can result in problems with coordinating activities, leading to costly and time-consuming delays, errors, and disputes concerning responsibility and financial liability.
Once a job has been delivered and accepted at the galvanizer’s plant, there is one point of responsibility for ensuring the material leaves the plant properly galvanized – the galvanizer.
Surface preparation for galvanizing typically consists of three steps: caustic cleaning, acid pickling, and fluxing.
Degreasing – A hot alkali solution is often used to remove organic contaminants such as dirt, paint markings, grease, and oil from the metal surface.
Epoxies, vinyls, asphalt, or welding slag must be removed before galvanizing by grit blasting, sand blasting, or other mechanical means.
Pickling – Scale and rust normally are removed from the steel surface by pickling in a dilute solution of hot sulfuric acid or ambient temperature hydrochloric acid.
Surface preparation also can be accomplished using abrasive cleaning as an alternative to or in conjunction with chemical cleaning. Abrasive cleaning is a process whereby sand, metallic shot, or grit is propelled against the steel material by air blasts or rapidly rotating wheels.
Fluxing – Fluxing is the final surface preparation step in the galvanizing process. Fluxing removes oxides and prevents further oxides from forming on the surface of the metal prior to galvanizing and promotes bonding of the zinc to the steel or iron surface.
Steel or iron materials are dipped in an aqueous solution of zinc ammonium chloride to apply the flux layer. The material is then thoroughly dried prior to immersion in liquid zinc.
Galvanizing
During the “galvanizing” process, the material is completely immersed in a bath consisting of a minimum of 98% pure liquid zinc. The bath chemistry is specified by ASTM International A123. The bath temperature is maintained at about 840 degrees Fahrenheit (449 degrees Celsius).
Fabricated items are immersed in the bath long enough to reach bath temperature. The articles are withdrawn slowly from the galvanizing bath and the excess zinc is removed by draining, vibrating, and/or centrifuging.
The chemical reactions that result in the formation and structure of the galvanized coating continue after the steel is withdrawn from the bath as long as the steel is near the bath temperature. The articles are cooled in either water or ambient air immediately after withdrawal from the bath.
Inspection
The first two properties of the hot-dip galvanized coating closely scrutinized after galvanizing are coating thickness and coating appearance. A variety of simply physical and laboratory tests may be performed to determine thickness, uniformity, adherence, and appearance.
Products are galvanized according to long-established, well-accepted, and approved standards of the American Society for Testing and Materials (ASTM) . These standards cover everything from the minimum required coating thicknesses for various galvanized items to the composition of the zinc metal used in the process.
*Life-Cycle Cost Calculator provided by the American Galvanizers Association.

